Filters
Question type
A) hydrogen bonds.
B) van der Waals interactions.
C) polar covalent bonds.
D) nonpolar covalent bonds.
E) ionic bonds.
F) None of the above
G) A) and B)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Question 52
Multiple Choice
Which of the following molecules contains the most polar covalent bond?
A) H₂
B) O₂
C) CO₂
D) H₂O
E) CH₄
F) None of the above
G) C) and E)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Question 53
Multiple Choice
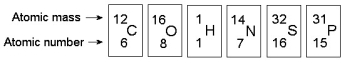 -In the figure above, how many electrons does nitrogen have in its valence shell?
-In the figure above, how many electrons does nitrogen have in its valence shell?
A) 2
B) 5
C) 7
D) 8
E) 14
F) A) and E)
G) None of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 81 - 83 of 83
Related Exams